The most common cancer among men after skin cancer, approximately 1 in 8 men will be diagnosed with prostate cancer in their lifetime. In 2022, the American Cancer Society (ACS) said prostate cancer diagnoses in the U.S. are nearly equivalent to breast cancer cases. Furthermore, African American men are nearly twice as likely to be diagnosed and die from prostate cancer compared to Caucasian men. ACS estimates that 268,490 cases will be diagnosed in the U.S. this year alone—and prostate cancer is being diagnosed at much earlier stages due to a simple blood test that measures a protein called prostate-specific antigen (PSA). The test can be ordered annually by a primary care physician or urologist. The key to successful treatment is early detection. When caught early, there are many treatment options for prostate cancer with high cure rates.

The Sonablate, a state-of-the art, non-invasive medical device that destroys tissue in the prostate using focused ultrasound
A HIGHLY FOCUSED TREATMENT
For the past 30 years, most prostate cancer patients have undergone whole-gland prostate cancer treatments with either surgery or radiation. Both treatments have high cure rates, but can cause permanent side effects such as erectile dysfunction (ED) and incontinence, which can be quite detrimental to a patient’s quality of life and can lead to depression. Just as breast cancer surgery moved from mastectomy to lumpectomy, prostate cancer treatment has started shifting from treating the entire prostate to only the cancerous portion of the gland. As a result of earlier diagnosis, made more precise with MRIs and novel genetic testing, many patients are candidates for focal treatments that only treat part of the prostate, allowing patients to avoid surgery and radiation, which often cause more harm than the cancer itself.
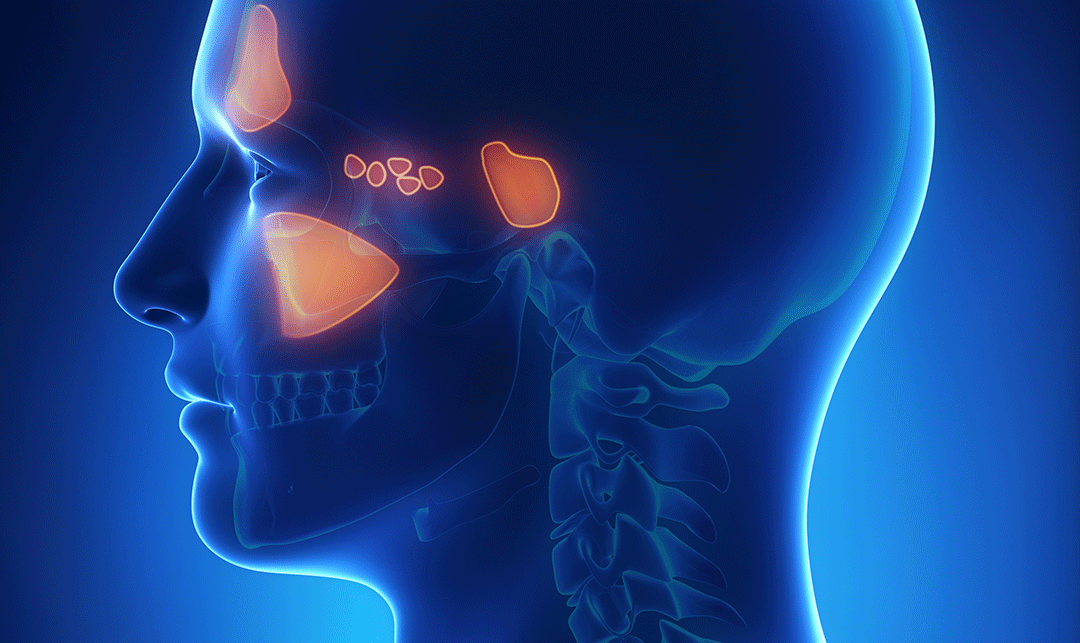
Often termed the “lumpectomy for men,” high-intensity focused ultrasound (HIFU) is a focal prostate cancer treatment that was cleared by the FDA in 2015. A noninvasive outpatient procedure that uses ultrasound energy to heat and destroy cancerous tissue in the prostate, HIFU is radiation free and bloodless, and studies show that the risk of side effects, such as ED and urinary incontinence, are significantly lower than surgery or radiation.
HIFU works in the same way that sunlight can be focused to burn a hole in a leaf. The procedure is performed under general anesthesia, so does not cause discomfort to the patient. In addition to the U.S., HIFU is approved in over 50 countries and has been used in Canada and Europe for nearly 20 years.
Since FDA clearance, HIFU has largely been a $25,000 self-pay treatment, as it takes time for Medicare and then commercial insurance to cover new procedures. Based on the preliminary 2023 outpatient treatment classifications announced by the Centers for Medicare & Medicaid Services, reimbursement will allow HIFU to be offered at significantly more hospitals and surgery centers with Medicare and insurance covering the facility and physician fees beginning next July. The lack of insurance coverage has significantly limited the number of patients that can afford treatment; however, widespread insurance coverage will speed physician adoption of HIFU.

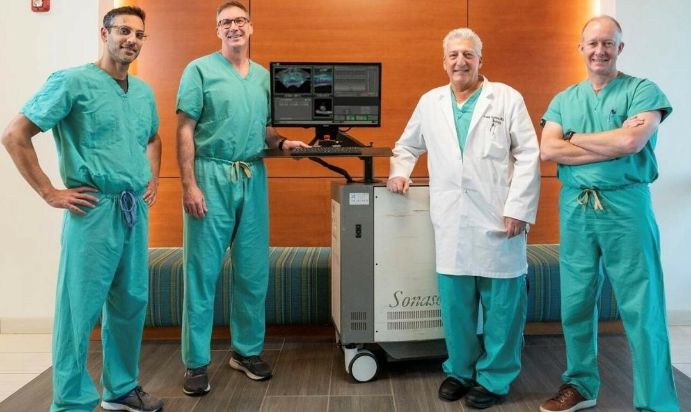
Dr. Eric Giesler with the Sonablate
EXPANDING THE REACH OF HIFU
I had the privilege of being part of a select few responsible for introducing HIFU into the U.S. After serving as a senior executive with Sonablate Corp., the designer and manufacturer of the HIFU system, I established HIFU Prostate Services (HPS) in late 2015 as a company focused on distributing the HIFU procedure to surgeons nationwide and providing treatment-related services. Throughout my 10 years with Sonablate Corp., I worked alongside many physicians who went on to become HPS affiliates just as they were becoming acquainted with the technology. Segue to 2022, and our company has built a network of more than 700 physicians—from single practitioners to large urology groups and academic universities across the U.S.
Insurance coverage will fuel our company’s growth and further expansion of HIFU as it enable physicians, outpatient facilities, and community hospitals to offer the breakthrough procedure.
An estimated 50,000 to 75,000 men per year select no treatment or watchful waiting due to the side effects and impact on quality of life from surgery and radiation. HIFU offers these patients and approximately 85% of those diagnosed with prostate cancer a curative option that will not impact their active lifestyles. The ideal HIFU patient will have capsule-confined prostate cancer (no metastasis), PSA of less than 20, Gleason score (a measurement of the tumor cells) of seven or less, and a prostate size of less than 40 grams.
HIFU represents a welcome treatment for those diagnosed with prostate cancer, one that effectively treats the cancer with similar cure rates to surgery while preserving quality of life, most notably a man’s ability to achieve erections and control their bladder. In addition, HIFU has a quick recovery time and patients are back to work in a couple of days and enjoying outdoor activities such as golf within a week of treatment.
Many industry experts see HIFU becoming one of the preferred treatment modalities for prostate cancer patients as insurance and physician adoption expand in 2023 and beyond. The continuing success of HIFU underscores how innovation and dedication can rise to the occasion—even in the competitive field of health services.
John Linn is founder and Chief Executive Officer of HIFU Prostate Services (HPS).